Impresszum | Copyright © 2026 Budapesti Műszaki és Gazdaságtudományi Egyetem
Selected publications |
2024 | 2023 | 2022 | 2021 | 2020 | 2019 |2018 | 2017 | 2016 | 2015 | 2014 | 2013 |
2024
|
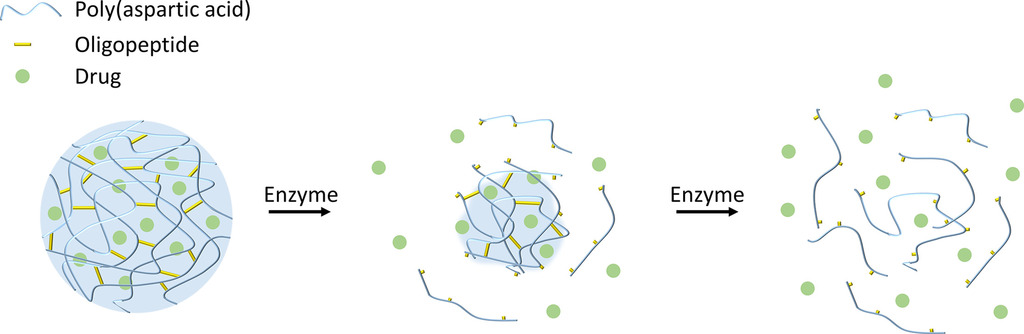
1. Stankovits G, Szayly K, Galata DL, Móczó J, Szilágyi A, Gyarmati B. Materials Today Bio 2024; 30: 101416
|
 |
2023
|
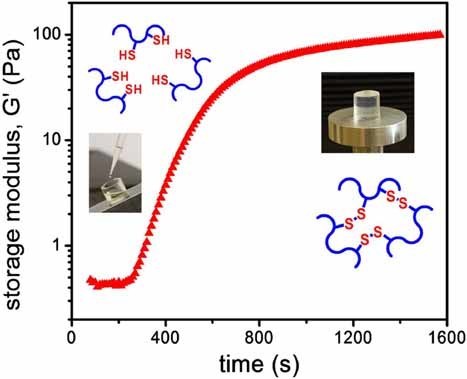
1. Stankovits G, Ábrahám Á, Kiss É, Varga Z, Misra A, Szilágyi A, Gyarmati B. International Journal of Biological Macromolecules 2023; 253: 126826
|
 |
|
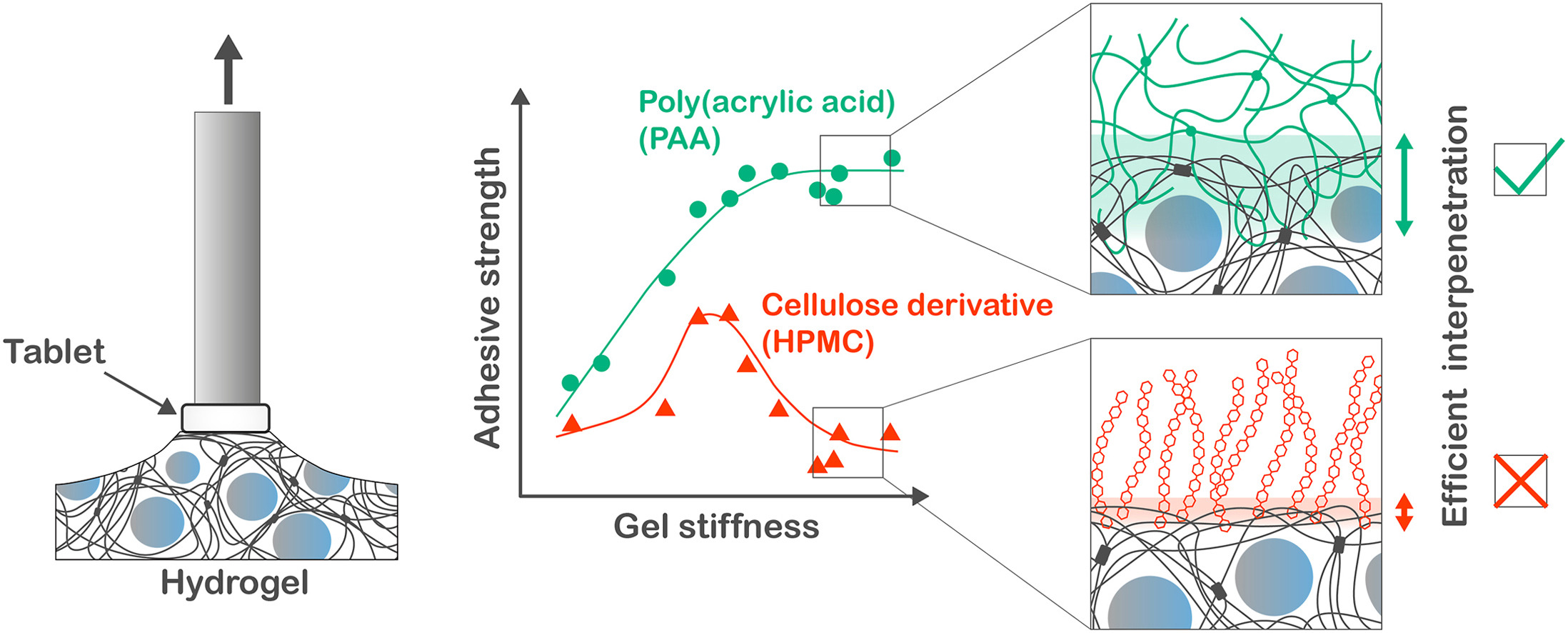
2. Szilágyi BÁ, Gyarmati B, Kiss EL, Budai-Szűcs M, Misra A, Csányi E, László K, Szilágyi A. Colloids and Surfaces B: Biointerfaces 2023; 225: 113254 |
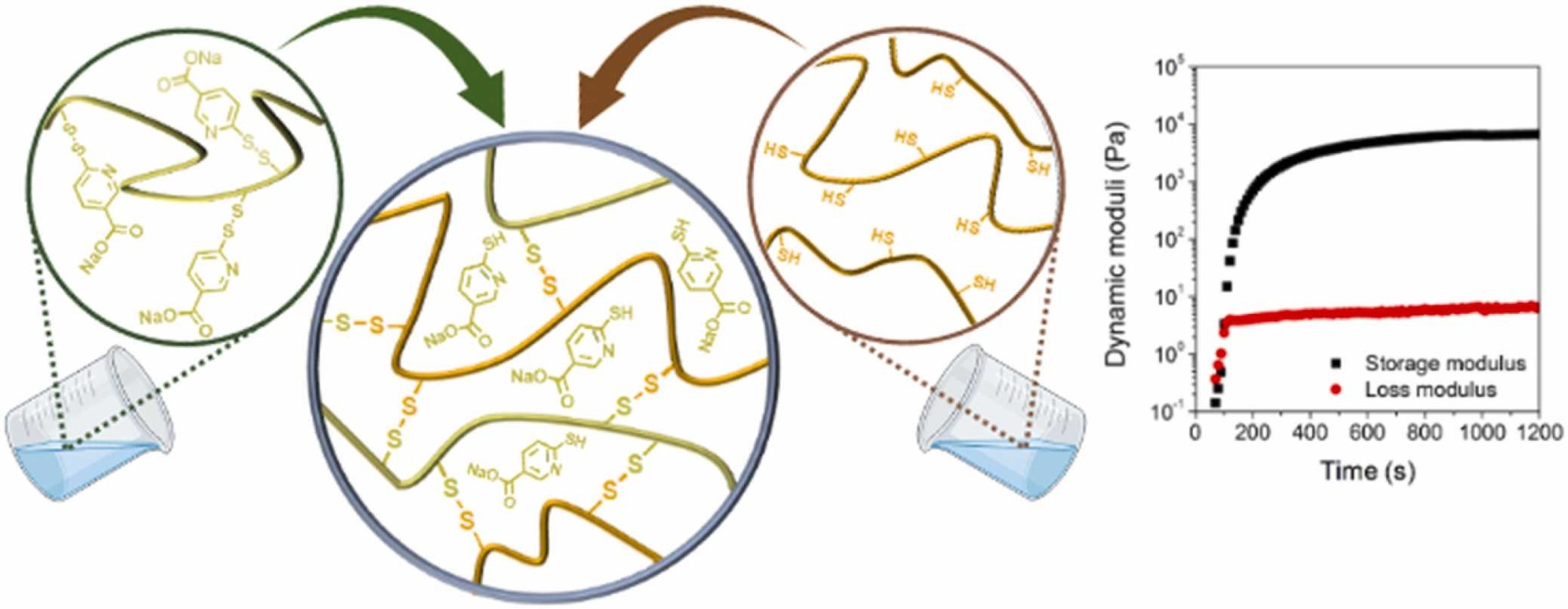 |
2022
|
1. Mammadova A, Gyarmati B, Sárdi K, Paudics A, Varga Z, Szilágyi A. Journal of Materials Chemistry B 2022; 10: 5946-5957 |
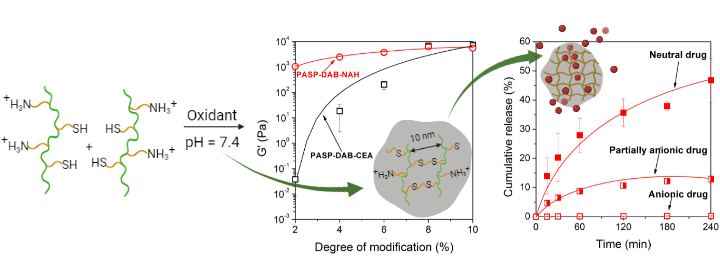 |
|
2. Gyarmati B, Stankovits G, Szilágyi BÁ, Galata DL, Gordon P, Szilágyi A. Colloids and Surfaces B: Biointerfaces 2022; 213: 112406 |
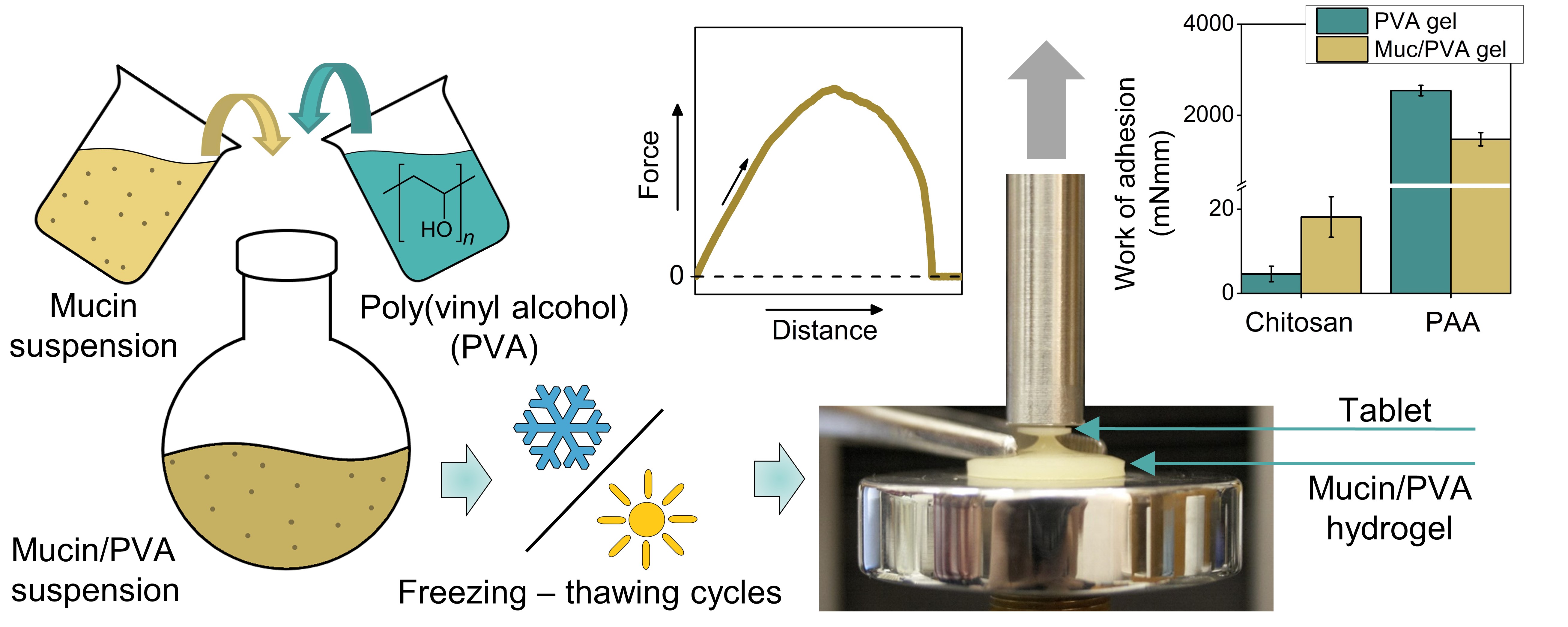 |
|
|
2021
|
|
|
1. Gyarmati B, Mammadova A, Barczikai D, Stankovits G, Misra A, Alavijeh SA, Varga Z, László K, Szilágyi A. Polymer Degradation and Stability 2021; 188: 109577 |
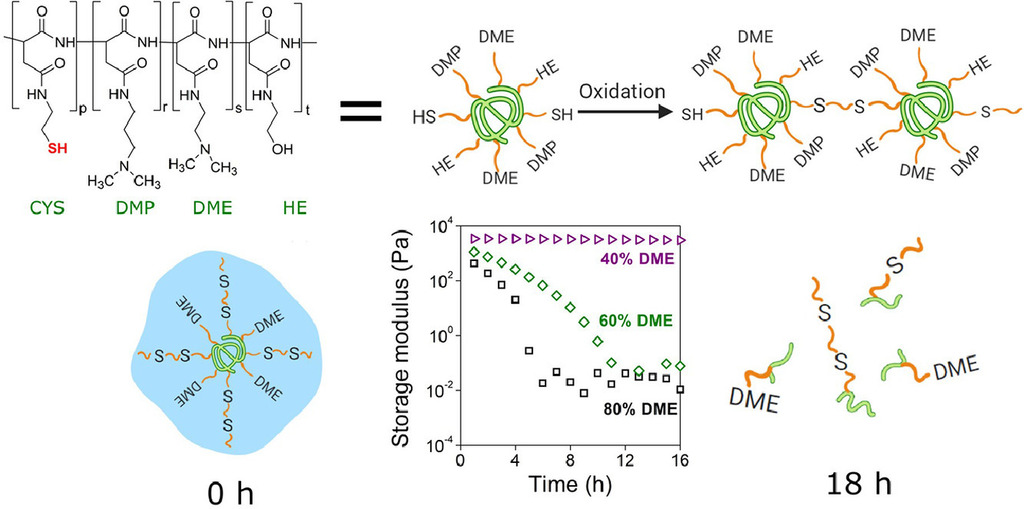 |
|
|
|
2. Gyarmati B, Mammadova A, Stankovits G, Barczikai D, Szilágyi A. Periodica Polytechnica Chemical Engineering 2021; 65:183-191 |
|
|
|
2020
|
|
|
1. Szilágyi BÁ, Mammadova A, Gyarmati B, Szilágyi A. Colloids and Surfaces B: Biointerfaces 2020; 194: 111219 |
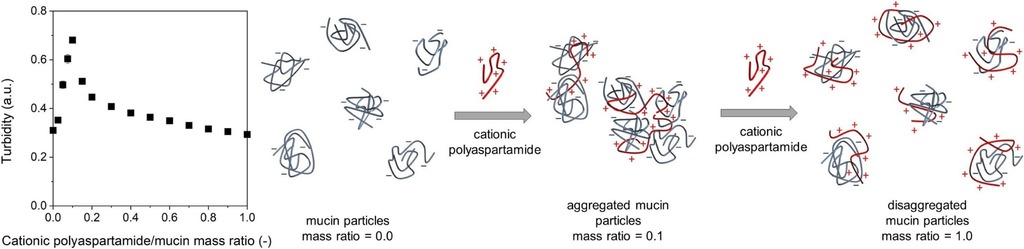 |
|
|
|
2. Németh C, Gyarmati B, Gacs J, Salakhieva DV, Molnár K, Abdullin T, László K, Szilágyi A. Fast dissolving nanofibrous matrices prepared by electrospinning of polyaspartamides European Polymer Journal 2020; 130: 109624 |
 |
|
|
2019
|
|
|
1. Decsi B, Krammer R, Hegedűs K, Ender F, Gyarmati B, Szilágyi A, Tőtős R, Katona G, Paizs C, Balogh GT, Poppe L, Balogh-Weiser D. Micromachines 2019; 10: 668 |
|
|
|
|
2. Gyarmati B, Pukánszky B. Natural polymers, bio-inspired and smart macromolecular materials European Polymer Journal 2019; 119: 393-399 |
|
|
|
|
3. Krisch E, Balogh-Weiser D, Klimko J, Gyarmati B, Laszlo K, Poppe L, Szilagyi A. Express Polymer Letters 2019; 13: 512-523 |
|
|
2018
|
|
|
1. Szilágyi BÁ, Némethy A, Magyar A, Szabó I, Bősze S, Gyarmati B, Szilágyi A. Amino acid based polymer hydrogel with enzymatically degradable cross-links Reactive and Functional Polymers 2018; 133: 21-28 |
|
|
|
|
2. Krammer O, Gyarmati B, Szilágyi A, Illés B, Bušek D, Dušek K. The effect of solder paste particle size on the thixotropic behaviour during stencil printing Journal of Materials Processing Technology 2018; 262:571-576 |
|
|
|
|
3. Balogh-Weiser D, Németh C, Ender F, Gyarmati B, Szilágyi A, Poppe L. Electrospun Nanofibers for Entrapment of Biomolecules Electrospinning Method Used to Create Functional Nanocomposites Films (2018, IntechOpen, ed. Tomasz Tański) |
|
|
|
|
4. Krisch E, Gyarmati B, Barczikai D, Lapeyre V, Szilágyi BÁ, Ravaine V, Szilágyi A. Poly(aspartic acid) hydrogels showing reversible volume change upon redox stimulus European Polymer Journal 2018; 105:459-468 |
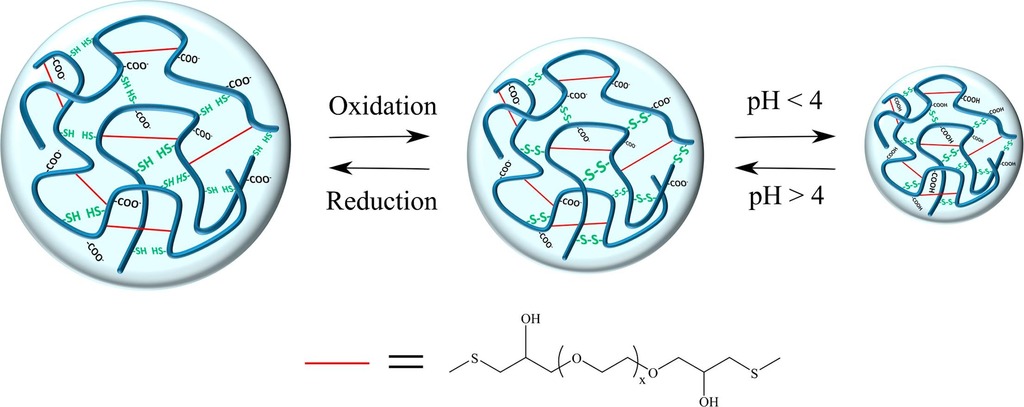 |
|
|
|
5. Orha L, Tukacs JM, Gyarmati B, Szilágyi A, Kollár L, Mika LT. ACS Sustainable Chemistry & Engineering 2018; 6: 5097-5104 |
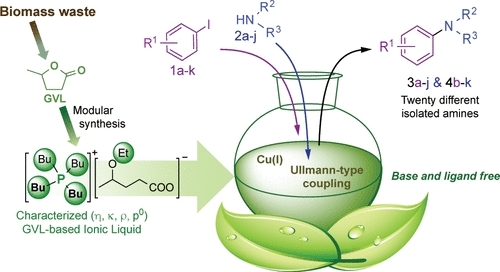 |
|
|
|
6. Budai-Szűcs M, Kiss EL, Szilágyi BÁ, Szilágyi A, Gyarmati B, Berkó S, Kovács A, Horvát G, Aigner Z, Soós J, Csányi E. Polymers 2018; 10: 199 |
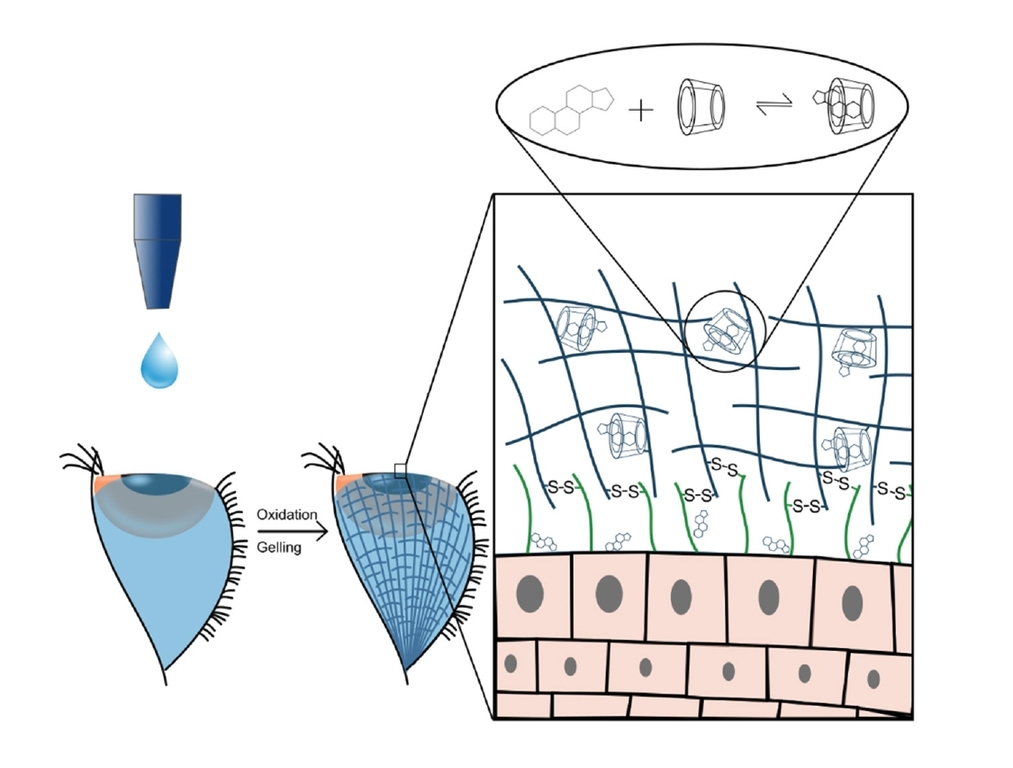 |
2017
|
|
|
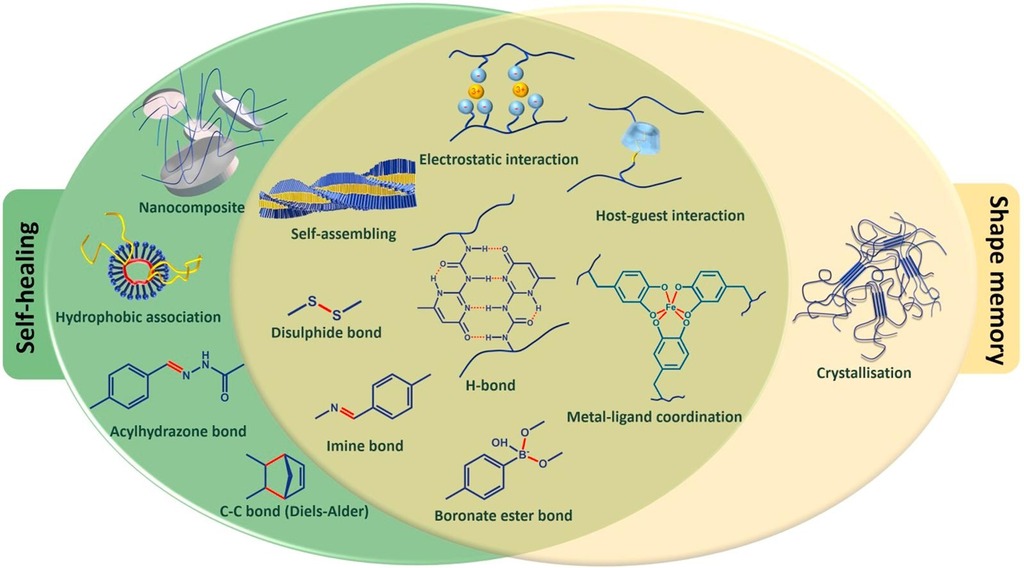
1. Gyarmati B, Szilágyi B, Szilágyi A. Reversible interactions in self-healing and shape memory hydrogels European Polymer Journal 2017; 93:642-669 |
 |
|
|
|
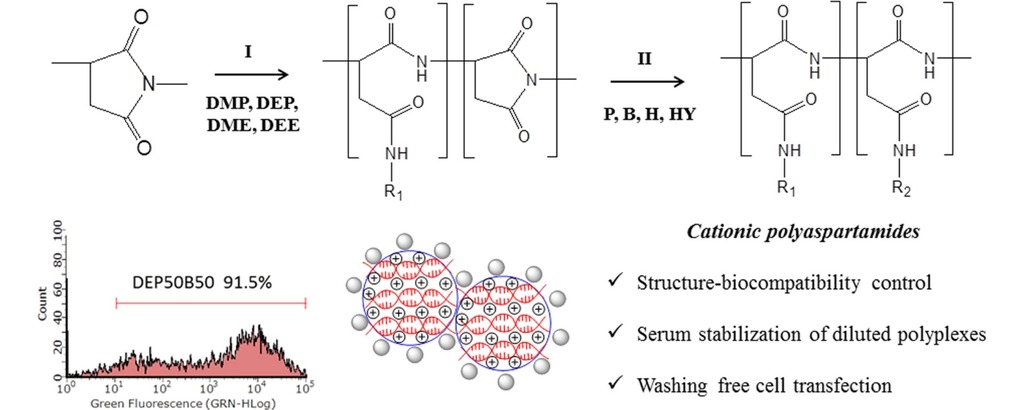
2. Németh Cs, Szabó D, Gyarmati B, Gerasimov A, Varfolomeev M, Abdullin T, László K, Szilágyi A. Effect of side groups on the properties of cationic polyaspartamides European Polymer Journal 2017; 93: 805-814 |
 |
|
|
|
3. Németh Cs, Gyarmati B, Abdullin T, László K, Szilágyi A. Poly(aspartic acid) with adjustable pH-dependent solubility Acta Biomaterialia 2017; 49: 486-494 |
 |
|
|
|
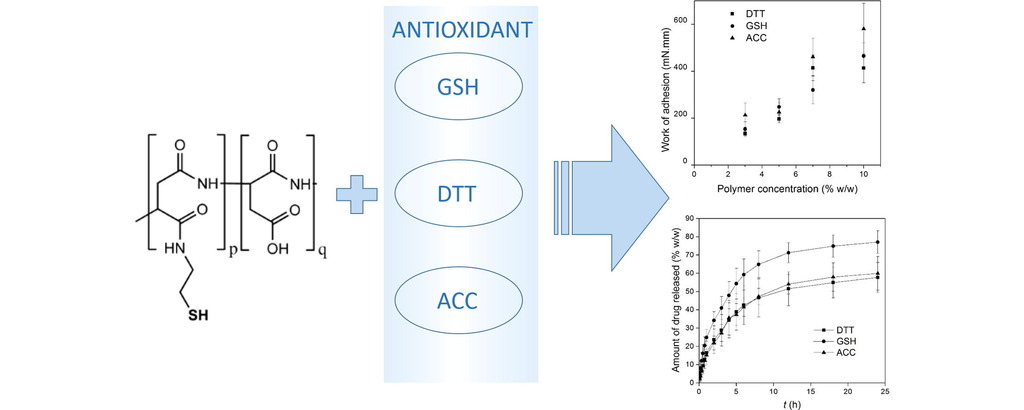
4. Budai-Szűcs M, Horvát G, Gyarmati B, Szilágyi BÁ, Szilágyi A, Berkó S, Ambrus R, Szabó-Révész P, Sandri G, Bonferoni MC, Caramella C, Csányi E. European Journal of Pharmaceutics and Biopharmaceutics 2017; 113: 178-187 |
 |
|
5. Krammer O, Gyarmati B, Szilágyi A, Storcz R, Jakab L, Illés B, Géczy A, Dušek K. Investigating the thixotropic behaviour of Type 4 solder paste during stencil printing Soldering & Surface Mount Technology 2017; 29: 10-14 |
|
|
6. Salakhieva D, Shevchenko V, Németh C, Gyarmati B, Szilágyi A, Abdullin T. International Journal of Pharmaceutics 2017; 517: 234-246 |
 |
|
7. Gyarmati B, Pukánszky B. Natural polymers and bio-inspired macromolecular materials European Polymer Journal 2017; 93: 612-617 |
|
|
|
|
8. Krisch E, Gyarmati B, Szilágyi A. Preparation of pH-responsive poly (aspartic acid) nanogels in inverse emulsion Periodica Polytechnica Chemical Engineering 2017; 61: 19-26 |
|
|
|
2016
|
|
|
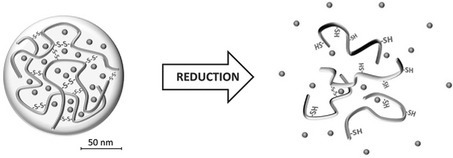
1. Krisch E, Messager L, Gyarmati B, Ravaine V, Szilágyi A. Redox- and pH-responsive nanogels based on thiolated poly(aspartic acid) Macromolecular Materials and Engineering 2016; 301: 260-266 |
 |
|
2. Müller P, Bere J, Fekete E, Móczó J, Nagy B, Kállay M, Gyarmati B, Pukánszky B. Interactions, structure and properties in PLA/plasticized starch blends Polymer 2016; 103: 9-18 |
 |
|
3. Budai-Szűcs M, Horvát G, Gyarmati B, Szilágyi BÁ, Szilagyi A, Csihi T, Berkó S, Szabó-Révész P, Mori M, Sandri G, Bonferoni MC, Caramella C, Csányi E. In vitro testing of thiolated poly(aspartic acid) from ophthalmic formulation aspects Drug Development and Industrial Pharmacy 2016; 42: 1241-1246 |
|
|
4. Budai-Szűcs M, Horvát G, Szilágy BÁ, Gyarmati B, Szilágyi A, Berkó S, Szabó-Révész P, Sandri G, Bonferoni MC, Caramella C, Soós J, Csányi AE. Journal of Ophthalmology 2016; 2647264 |
|
2015
|
|
|
1. Gyarmati B, Mészár EZ, Kiss L, Deli M, László K, Szilágyi A. Supermacroporous chemically cross-linked poly(aspartic acid) hydrogels Acta Biomaterialia 2015; 22: 32-38. |
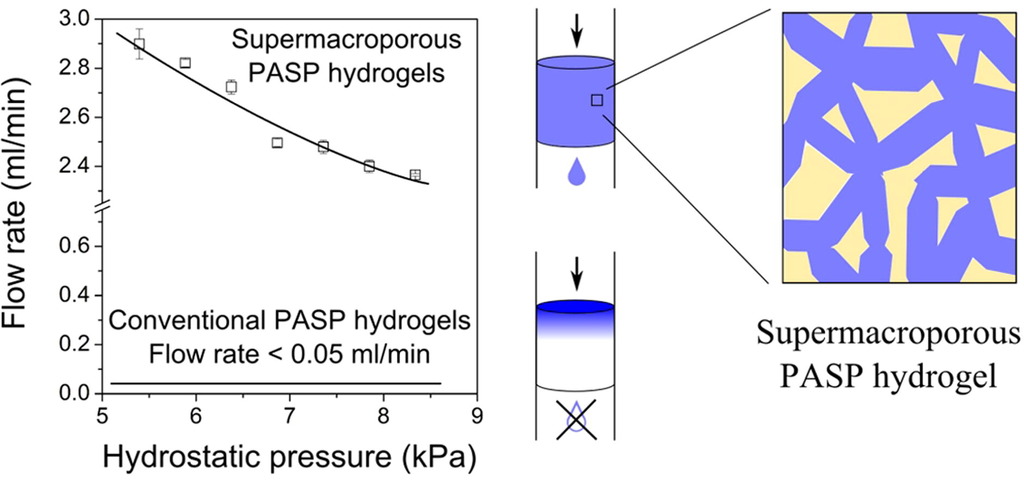 |
|
|
|
|
2. Solti K, Gyarmati B, Szilágyi A. Szívós polimer hidrogélek Műanyag- és gumiipari évkönyv 2015;13:100-108. |
|
|
|
|
|
3. Horvát G, Gyarmati B, Berkó Sz, Szabó-Révész P, Szilágyi BÁ, Szilágyi A, Soós J, Sandri G, Bonferoni MC, Rossi S, Ferrari F, Caramella C, Csányi E, Budai-Szűcs M. Thiolated poly(aspartic acid) as potential in situ gelling, ocular mucoadhesive drug delivery system European Journal of Pharmaceutical Sciences 2015; 67: 1-11 |
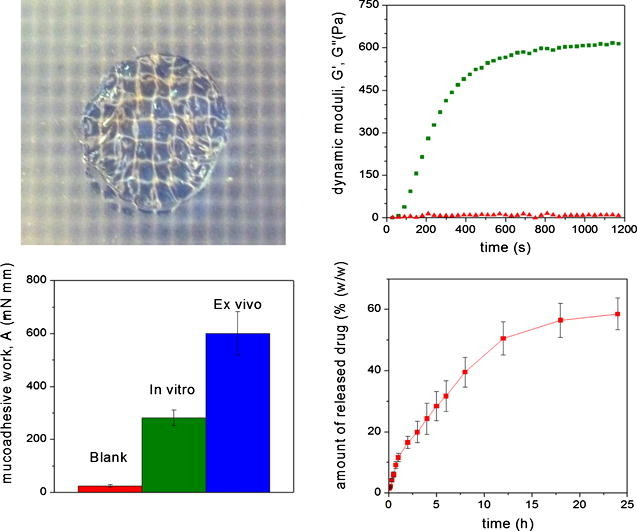 |
|
|
|
4. Gyarmati B, Hegyesi N, Pukánszky B, Szilágyi A. Express Polym. Lett. 2015;9:154-164. |
|
|
|
|
5. Horvát G, Budai-Szűcs M, Berkó S, Révész P, Gyarmati BS, Szilágyi BÁ, Szilágyi AF, Csányi E. Egy új típusú polimer, az in situ gélesedő tiolált poliaszparaginsav alkalmazása szemészeti gyógyszerhordozóként Acta Pharmaceutica Hungarica 2015; 85: 115-122 |
|
|
|
|
2014
|
|
|
1. Gyarmati B, Némethy Á, Szilágyi A. Reversible response of poly(aspartic acid) hydrogels to external redox and pH stimuli RSC Advances 2014;4:8764-8771 |
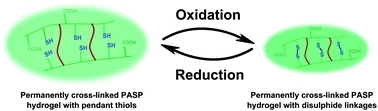 |
|
2. Gyarmati B, Krisch E, Szilágyi A. In situ oxidation-induced gelation of poly(aspartic acid) thiomers Reactive and Functional Polymers 2014; 84: 29-36 |
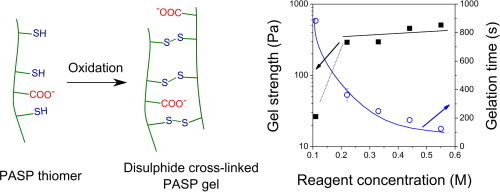 |
|
3. Horváth Z, Gyarmati B, Menyhárd A, Doshev P, Gahleitner M, Varga J, Pukánszky B. Drug development and industrial pharmacy 2016; 42: 1241-1246 |
|
|
4. Gyarmati B, Szilágyi A. Preface for papers presented at AMSALS 2012 Periodica Polytechnica Chemical Engineering 2014;58:47-48 |
|
2013
|
|
|
1. Némethy Á, Solti K, Kiss L, Gyarmati B, Deli MA, Csányi E, Szilágyi A. pH-and temperature-responsive poly(aspartic acid)-l-poly(N-isopropylacrylamide) conetwork hydrogel European Polymer Journal 2013; 49: 2392-2403 |
 |
|
2. Gyarmati B, Némethy Á, Szilágyi A. European Polymer Journal 2013; 49: 1268-1286 |
 |
|
3. Gyarmati B, Vajna B, Némethy Á, László K, Szilágyi A. Macromolecular Bioscience 2013;13: 633-640 |
|